View letters related to this article
ABSTRACT
Calcium hydroxide, widely used in endodontic treatment, is a strong base that may cause irreversible injury to vital tissue that comes into contact with this substance. We present the first case of a dentist who accidentally splashed endodontic calcium hydroxide into her own eye. After washing with copious amounts of water for several minutes, she was treated in the hospital within 30 minutes of the accident. Because of the burning caused by the base solution, the dentist lost vision in the affected eye. She returned to the hospital several times for treatment of a corneal abscess and corneal fungal infection. She had the keloid that formed between the eyeball and eyelid removed 3 times. Calcium hydroxide can cause blindness when it comes into contact with the eye. Clinicians should take adequate precautions to prevent this serious complication. In case of an accident, it is important to wash the eye efficiently.
Calcium hydroxideis widely used as an intracanal medication in root canal treatments. The most common and effective type of calcium hydroxide is in an aqueous suspension. It kills a wide spectrum of bacteria and remains active for a long time. The antibacterial effect of calcium hydroxide is due directly to its high pH (12–13).1-4 However, tissue damage occurs when calcium hydroxide is applied beyond the root canal space. De Bruyne and others,5 and Bramante and others6 described cases of overextending a calcium hydroxide intracanal dressing into the periradicular and soft tissue after iatrogenic perforation of maxillary incisors. The result was severe necrosis of gingival and oral mucosa. Sharma and others7 described 2 cases in which calcium hydroxide was injected into an artery through the root canal system of maxillary canals. In both cases, the insult resulted in severe clinical signs, including necrosis of the gingival mucosa, oral mucosa and the skin. Ahlgren and others8 reported inferior alveolar nerve paresthesia caused by calcium hydroxide paste introduced into the mandibular canal. However, Fava9 described a case of calcium hydroxide overfilling in the maxillary sinus that was beneficial: 3 months after the incident, the patient was free of symptoms, although a radiographic mass was visible in the sinus. Recently, another study10 described a case of necrosis observed on the left infraorbital area of the skin and concomitant hypoesthesia of the infraorbital nerve and mental nerve (embolia cutis medicamentosa) after calcium hydroxide was injected during endodontic treatment.
Here, we present a case of a calcium hydroxide intracanal dressing accidentally splashed into the eye of a dentist during root canal therapy that resulted in the loss of all sight in the injured eye. To our knowledge, no similar case has been previously reported.
Case Report
A dentist accidentally splashed calcium hydroxide into her own eye while applying an aseptic endodontic treatment to a second mandibular premolar. When the dentist tried to apply a calcium hydroxide intracanal dressing (Calasept, Scania Dental, Sweden) directly from a syringe with a needle, she found that the needle was blocked. To examine the patency of the needle closely, she removed her protective glasses and pressed the syringe plunger. The preparation then ejected from the needle directly into her left eye, causing an instant, strong, burning pain. The dentist immediately proceeded to rinse her eye with tap water for several minutes. During this procedure, she felt a sharp, unpleasant, tearing chest pain that radiated out to both shoulders and the mandible. With help, she went to the hospital and underwent treatment about 30 minutes after the incident. The treatment at the hospital included cleaning the remaining calcium hydroxide from the conjunctival sac and its fornices, prolonged rinsing of the conjunctival sac with 0.9% sodium chloride, administering the patient’s own blood under the conjunctiva, and neutralizing the calcium hydroxide with sodium edetate, according to the treatment protocol.
The ophthalmic examination revealed a lack of tear secretion, shortening of the conjunctival sack (adhesion between the palpebral conjunctiva and the ocular conjunctiva), swollen palpebral and ocular conjunctivas, and burned tissue. The cornea was deeply burned, appearing opaque like porcelain. Corneal fluorescein staining was positive, indicating damage to the cornea. Other sections of the eye could not be examined clinically because of the lack of corneal translucency.
The symptoms of chest pain suggested a diagnosis of myocardial infarction, which was confirmed by cardiac diagnostics. Both cardiac and ophalmologic treatments commenced concurrently. Intensive conservative pharmacological treatment was applied to the eye; however, because of the chemical burn, oculopalpebral adhesions had formed, and the conjunctival sac had become shallow because of adhesions between the ocular and palpebral conjunctivas. The dentist lost vision in the injured eye because of the burning. She returned to the hospital several times for treatment of a corneal abscess and corneal fungal infection (Fig. 1). In addition, she had a keloid that formed between the eyeball and eyelid removed 3 times, and plastic surgery on the conjunctiva 3 times.
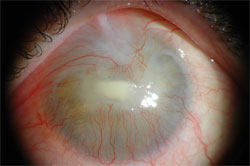 Figure 1: Photograph of a corneal ulceration in the process of healing. Located in the centre of the cornea, the ulceration has affected the ingrowing vessels from the limbus region that form a seam toward the centre of the cornea. The cornea is entirely opaque.
Figure 1: Photograph of a corneal ulceration in the process of healing. Located in the centre of the cornea, the ulceration has affected the ingrowing vessels from the limbus region that form a seam toward the centre of the cornea. The cornea is entirely opaque.
Discussion
Chemical eye injuries caused by exposure to acids and alkalis (bases) are the most common type of ocular burn. Alkali burns are typically more serious than acid burns because the alkali can rapidly penetrate the cornea into the anterior chamber of the eye (5–15 minutes) and damage the iris, ciliary body, lens and trabecular network.11-13 Alkali damage includes saponification of cell membranes, cell death and disruption of the extracellular matrix. Alkalis can continue destroying tissues within the eye for several days. Penetration rates differ with different types of alkalis. One of the most rapidly penetrating alkalis is ammonium hydroxide, followed by sodium hydroxide, potassium hydroxide, calcium hydroxide and sodium hypochlorite. Although calcium hydroxide penetrates more slowly than several other alkalis, it quickly saponifies membranes, and the newly created soap particles tend to gather in the fornices that provide a reservoir for ongoing injury.14,15
In this case report, a dentist lost total sight in one eye after splashing calcium hydroxide into her eye. To our knowledge, this is the first case of a chemical injury to the eye caused by endodontic calcium hydroxide. However, Ingram16 reported a case of a corneal burn caused by splashing a 5.25% sodium hypochlorite solution into the eye of a patient during endodontic therapy. In that case, the hypochlorite caused only slight edema of the conjunctiva and loss of epithelial cells in the cornea. Two days after the accident, the patient was asymptomatic and seemed completely healed. However, the dentist in this case report had severe damage and lost all sight in the affected eye. This was probably due to the ineffective removal of the calcium hydroxide at the site of injury, likely because the calcium hydroxide was trapped in saponified tissues, which made it difficult to remove from the recesses of the eye. For effective removal, the eyelid should be flipped inside-out, but this was done too late for our patient. Mechanical removal of the particles in her eye was done in the hospital 30 minutes after the injury.
Alkali substances can penetrate the cornea to the anterior chamber of the eye and cause irreversible destruction of the deeper structures of the eye within several minutes. Training health care personnel in the skills of turning the eyelid inside-out and thorough eye washing on site within the first few minutes of an accident is of prime importance. According to the latest guidelines, the eye should be washed for several minutes with saline or tap water; specialist help and treatment should be sought immediately after washing is completed.
Conclusion
This case report emphasizes the importance of taking special care with calcium hydroxide, a commonly used endodontic treatment, to avoid eye contamination because it can cause blindness. Both the patient and the care provider should wear eye protection, such as glasses or a dental safety face mask. If calcium hydroxide is splashed into the eye, the eyelid flip should be done immediately with copious washing, followed by prompt ophthalmologic consultation.
THE AUTHORS
References
- Duarte MA, Midena RZ, Zeferino MA, Vivan RR, Weckwerth PH, Dos Santos F, et al. Evaluation of pH and calcium ion release of calcium hydroxide pastes containing different substances. J Endod. 2009;35(9):1274-7. Epub 2009 Jul 2.
- Ballal NV, Shavi GV, Kumar R, Kundabala M, Bhat KS. In vitro sustained release of calcium ions and pH maintenance from different vehicles containing calcium hydroxide. J Endod. 2010;36(5):862-6. Epub 2010 Feb 21.
- Delgado RJ, Gasparoto TH, Sipert CR, Pinheiro CR, Moraes IG, Garcia RB, et al. Antimicrobial effects of calcium hydroxide and chlorhexidine on Enterococcus faecalis. J Endod. 2010;36(8):1389-93. Epub 2010 Jun 19.
- Asgary S, Nosrat A, Seifi A. Management of inflammatory external root resorption by using calcium-enriched mixture cement: a case report.J Endod. 2011;37(3):411-3. Epub 2010 Dec 30.
- De Bruyne MA, De Moor RJ, Raes FM. Necrosis of the gingiva caused by calcium hydroxide: a case report. Int Endod J. 2000;33(1):67-71.
- Bramante CM, Luna-Cruz SM, Siepert CR, Bernadineli N, Garcia RB, de Moraes IG, et al. Alveolar mucosa necrosis induced by utilisation of calcium hydroxide as a root canal dressing. Int Dent J. 2008;58(2):81-5.
- Sharma S, Hackett R, Webb R, Macpherson D. Wilson A. Severe tissue necrosis following intra-arterial injection of endodontic calcium hydroxide: a case series. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105(5):666-9.
- Ahlgren F, Johannessen ACH, Hellem S. Displaced calcium hydroxide paste causing inferior alveolar nerve paraesthesia: report of case. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;96(6):734-7.
- Fava R. Calcium hydroxide paste in the maxillary sinus: a case report. Int Endod J. 1993;26(5):306-10.
- Wilbrand JF, Wilbrand M, Schaaf H, Howaldt HP, Malik CY, Streckbein P. Embolia cutis medicamentosa (Nicolau syndrome) after endodontic treatment: a case report. J Endod. 2011;37(6):875-7. Epub 2011 Mar 3.
- Adepoju FG, Adeboye A, Adigun IA. Chemical eye injuries: presentation and management difficulties. Ann Afr Med. 2007;6(1):7-11.
- Kuckelkom R, Schrage N, Keller G, Redbrake C. Emergency treatment of chemical and thermal eye burns. Acta Ophthalmol Scand. 2002;80(1):4-10.
- Schrage NF, Langefeld S, Zschocke J, Kuckelkom R, Redbrake C, Reim M. Eye burns: an emergency and continuing problem. Burns. 2000;26(8):689-99.
- Agarwal T, Vajpayee RB, Sharma N, Tandon R. Severe ocular injury resulting from chuna packets. Ophthalmology. 2006;113(6):961.e1. Epub 2006 Apr 17.
- Schmidt SM, Schmidt CJ, Adler M, Rahmani B. Corneal injury due to calcium hydroxide containing food preparation product (“cal”). Pediatr Emerg Care. 2008;24(7):468-70.
- Ingram TA 3rd. Response of the human eye to accidental exposure to sodium hypochlorite. J Endod. 1990;16(5):235-8.
